Medicine for the money, not for the ‘few’ lives it’d save
French drugmaker won’t reopen cholera vaccine manufacturing despite global rush to save lives.
Kiri Rupiah
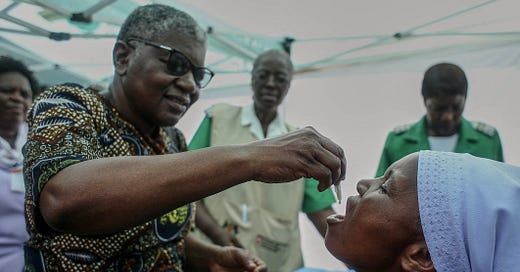
With at least 17 countries reporting outbreaks in the past year, the world has used up the available stockpile of cholera vaccines and new ones are not being produced fast enough.
Its manufacture is typically demanddriven and currently only one company, EuBiologics in South Korea, makes the vaccine. A French company, Sanofi, whose India subsidiary used to make about 15% of the world’s annual supply, pulled out of the supply chain in 2020. EuBiologics makes about 50-million doses a year but right now, the countries fighting outbreaks need at least 76-million doses.
Profit-driven pharmaceutical manufacturers don’t invest in the vaccine because there is little money to be made from it – the disease mostly affects poor people and isn’t included in the routine vaccinations countries and global vaccine alliances buy in bulk each year.
Until recently, the vaccine’s South Korean maker kept up with demand because outbreaks were rare and isolated. But extreme climate events and breakdowns in political order have driven surges across several regions and countries, including Malawi, Zimbabwe, Haiti, Yemen, the Democratic Republic of the Congo, Comoros, and Ethiopia. A warmer climate has also extended the lifespan of the cholera bacteria, meaning that outbreaks last longer.
Southern Africa is battling the deadliest outbreak of cholera in at least 10 years. Worldwide, according to the data from the European Centre for Disease Prevention and Control, there have been at least 150,866 cholera cases in the past year and over 1,700 have died.
In April, the WHO approved Euvichol-S, a version of the EuBiologics vaccine that uses fewer ingredients and is cheaper and faster to make.
Two companies in India and South Africa, Biological-E and Biovac respectively, are slated to begin manufacturing the vaccine but likely won’t put any on the market until the end of next year. The French company, which has already built capacity, continues to stay out of it despite pleas from the WHO.